|
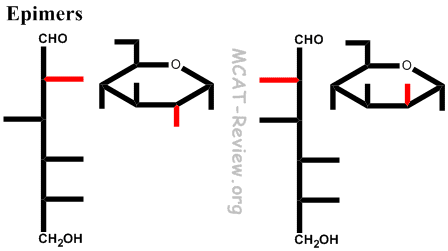
(Figure-10).įigure-10- Alpha and beta anomers of glucose In alpha anomer, the orientation of the OH group is towards the right side whereas, in the beta anomer, it is towards the left side. The carbon about which this rotation occurs is called Anomeric carbon and the two stereoisomers are called Anomers. The ring can open and reclose allowing the rotation to occur around the carbon bearing the reactive carbonyl group yielding two possible configurations- α and β of the hemiacetal and hemiketal. Similarly, the ring structure of a ketose is a hemiketal. The ring structure of an aldose is a hemiacetal since it is formed by the combination of an aldehyde (C1) and an alcohol group (Mostly C5). In a biological system, the monosaccharides tend to exist in a ring form. Glyceraldehyde and Dihydroxyacetone, Ribose and Ribulose are other examples of aldose -ketose isomers.įructose has the same molecular formula as glucose but it differs in its structural formula, since there is a potential keto group in position 2, the anomeric carbon of fructose (Figure-9), whereas there is a potential aldehyde group in position 1, the anomeric carbon of glucose. Mannose and Galactose are not epimers of each other as they differ in configuration around 2 carbon atoms (figure-8).įigure-7- Glucose and galactose are C4 epimersįigure-8- Relationship of glucose, galactose, and mannoseĬompounds with the same molecular formula but differing in nature of the functional group (aldehyde or keto) are aldose ketose isomers.Įxamples- Fructose and Glucose are aldose ketose isomers.įigure-9- Aldose ketose isomers (D-Glucose and D-Fructose) Biologically, the most important epimers of glucose are mannose and galactose, formed by epimerization at carbons 2(figure-6) and 4(figure-7), respectively.įigure-6- Glucose and Mannose are C2 epimers. In hexoses, isomers differing as a result of variations in the configuration of the -OH and -H on carbon atoms 2, 3, and 4 are known as epimers. The compounds with the same molecular formula, but differing in the spatial configuration of the attached groups around a single carbon atom only are called epimers. In solution, glucose is dextrorotatory, and glucose solutions are sometimes known as dextrose.įigure-5- Rotation of polarized light by an optically active solution The measurement of optical activity is done by an instrument called Polarimeter.įor example, the naturally occurring form of fructose is the D (–) isomer. Measurement of optical activity in chiral or asymmetric molecules using plane-polarized light is called Polarimetry. The direction of rotation of polarized light is independent of the stereochemistry of the sugar, so it may be designated D (–), D (+), L (–), or L (+) – figure-5 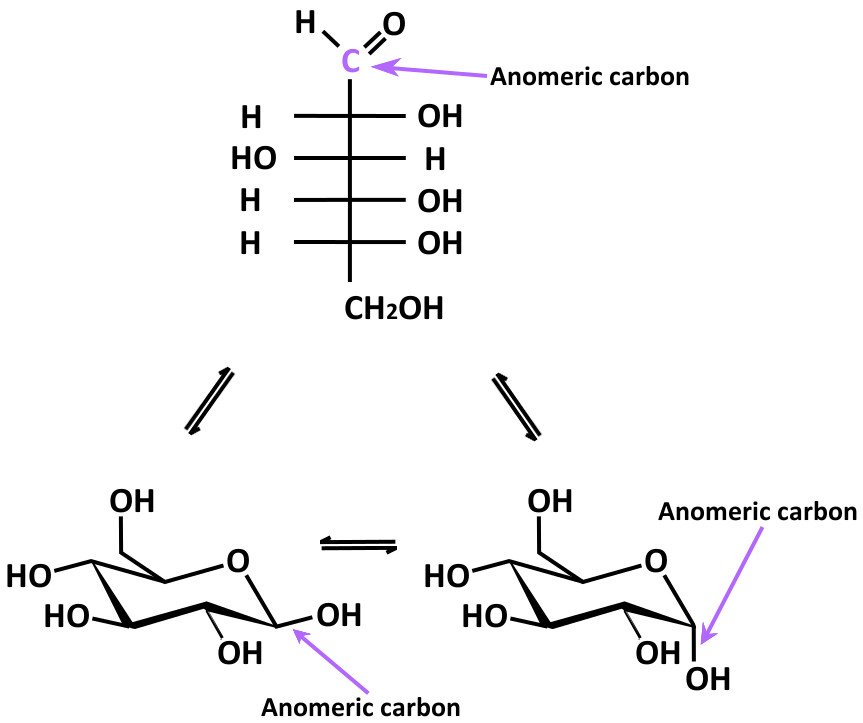
When a beam of plane-polarized light is passed through a solution of an optical isomer, it rotates either to the right, dextrorotatory (+), or to the left, levorotatory (–). L-Arabinose and L-fucose are found in glycoproteins.Ģ) Optical Isomerism- The presence of asymmetric carbon atoms also confers optical activity on the compound. Some sugars naturally occur in the L form e.g.D-Glucose, D-mannose, and D -galactose are abundant six-carbon aldoses.D-Ribose, the carbohydrate component of RNA, is a five-carbon aldose. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
